Glycolysis, Krebs, and the Electron Transport Chain (Explained for Humans)
A Glycolysis, Krebs Cycle, and ETC survival guide.
This post accompanies a YouTube video I created for my Master’s in Physiology program at the University of Louisville. The video was produced for PHZB-621-01-4262: Exercise and Inactivity Physiology (Spring 2026), with the assignment being simple in theory: explain glycolysis, the Krebs cycle, and the electron transport chain.
In practice, that meant translating one of the most memorization-heavy sections of physiology into something watchable, intuitive, and hopefully not miserable.
If you’ve ever stared at a metabolic pathway and thought, “I understand none of this, but I will memorize it anyway,” this video is for you.
Rather than reciting reactions step by step, I focused on the story of cellular respiration. Where energy actually comes from. Why electrons matter. What ATP production is really doing. And how glycolysis, the Krebs cycle, and the electron transport chain fit together as a system instead of three disconnected charts.
This video reflects how I learn best: talking things through, making analogies, and occasionally admitting when something is confusing before it becomes clear. It’s also a good snapshot of how I approach teaching and communication more broadly, especially in technical or medical contexts.
If you’re a student, I hope this helps.
If you’re reviewing physiology, I hope it refreshes.
And if you’re just curious how your cells turn food into usable energy, welcome.
▶️ Watch the video below.
The Process of Making This Video.
The goal of this video was twofold: to learn cellular respiration deeply enough to explain it, and to do so within a five-minute time limit.
To prepare, I used Excalidraw to sketch the pathways involved in glycolysis, the Krebs cycle, and the electron transport chain. The drawings were not intended to be polished instructional graphics. They were thinking tools. Sketching the pathways by hand forced me to confront gaps in my understanding and clarify how energy and electrons move through the system.
I used iMovie to edit the final video, adjusting pacing, trimming pauses, and increasing speed where necessary to stay under the five-minute constraint. In practice, many pre-medical and medical students already watch lecture material at increased playback speed, so the pacing felt reasonable to me. That said, I recognize that for someone encountering this material for the first time, the video may feel dense.
For that reason, I am unpacking the content more slowly on this page, using screenshots from the original drawing to walk through the concepts step by step. You can view the full working diagram here.
Glycolysis
Before discussing the Krebs cycle or the electron transport chain, it is important to start with glycolysis and its role in cellular respiration.
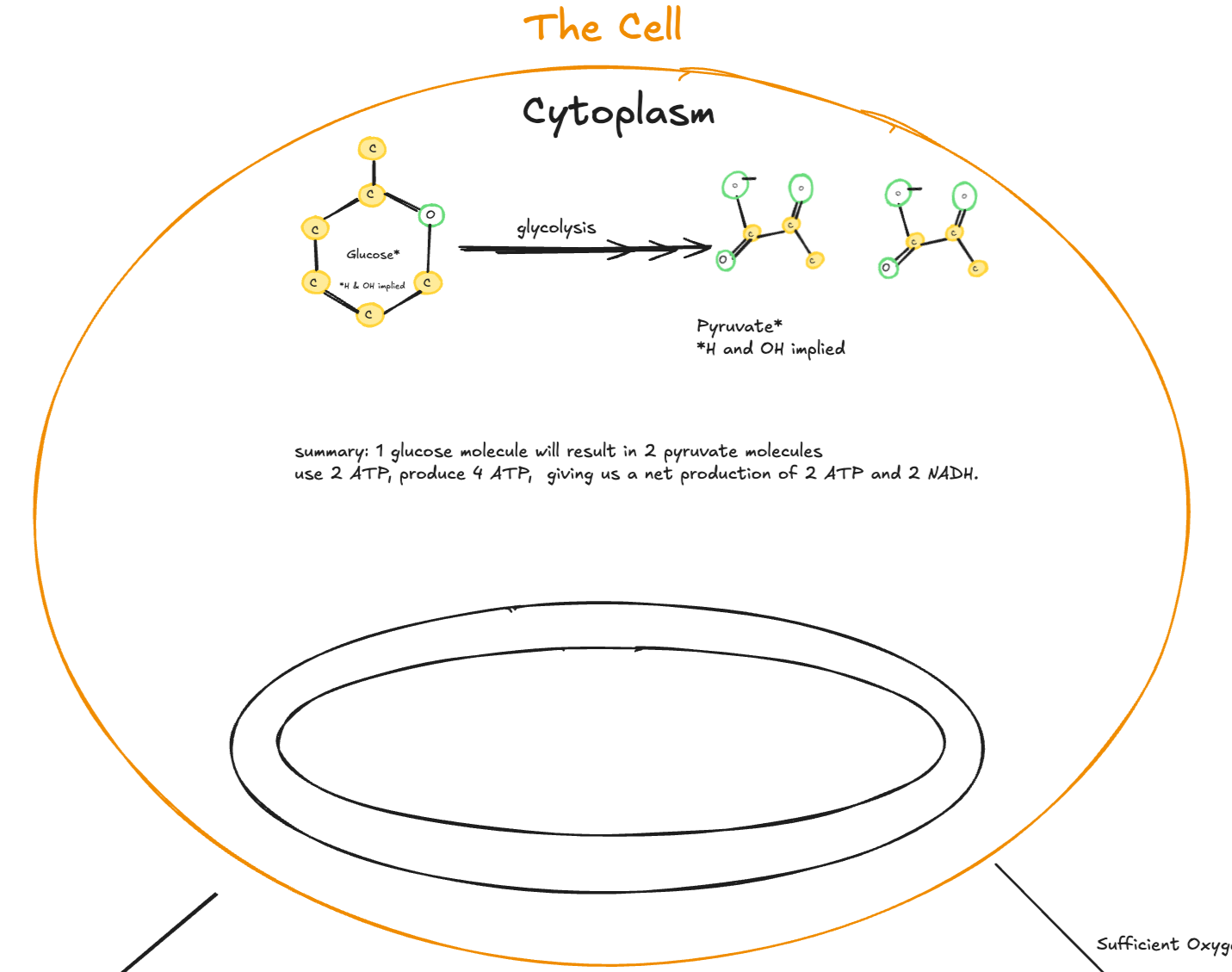
Digitally drawn overview of glycolysis in the cytoplasm, showing one glucose molecule converted into two pyruvate molecules, with a summary indicating ATP investment, ATP production, and net ATP and NADH yield.
At its most basic level, glycolysis is the breakdown of glucose. The name itself reflects this: glyco referring to glucose, and lysis meaning to split or break down.
In the body, glucose is transported from the bloodstream into the cell, where it enters as a six-carbon molecule. Glycolysis is a ten-step series of chemical reactions that converts this six-carbon structure into two three-carbon molecules. This process does not require oxygen and serves as the first stage of cellular respiration, setting the stage for further energy extraction.
The First Step of Glycolysis
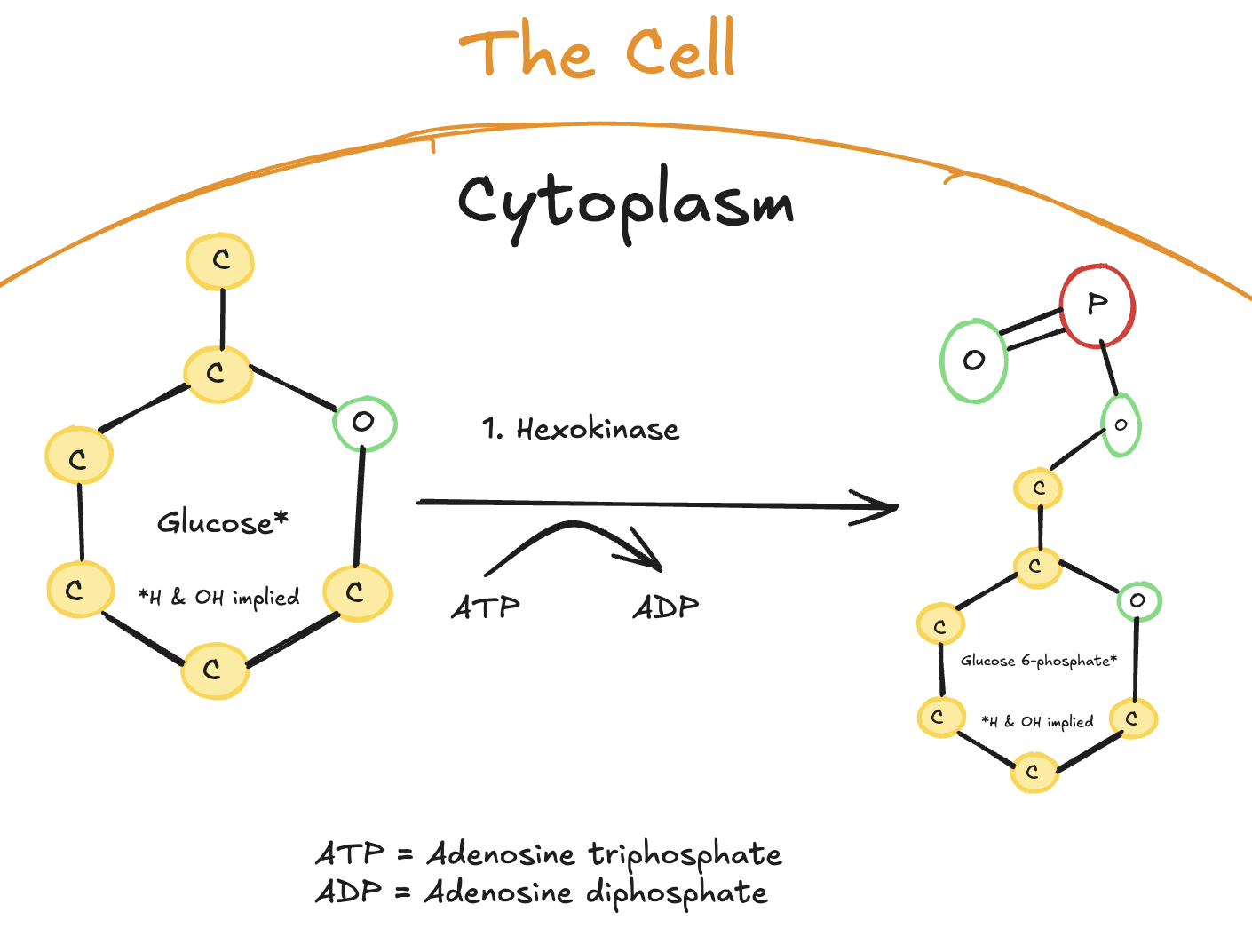
The first step of glycolysis is the phosphorylation of glucose.
Digitally drawn diagram of the first step of glycolysis in the cytoplasm, depicting a six-carbon glucose molecule converted to glucose-6-phosphate by hexokinase, with ATP donating a phosphate group and becoming ADP.
Once glucose enters the cell, it is immediately phosphorylated by the enzyme hexokinase (or glucokinase in the liver). This reaction uses one molecule of ATP to add a phosphate group to glucose, forming glucose-6-phosphate.
This step serves two important purposes. First, it traps glucose inside the cell, because the phosphorylated form cannot easily cross the cell membrane. Second, it commits glucose to metabolism, ensuring that it is processed through glycolysis rather than diffusing back into the bloodstream.
Key Takeaways
Enzyme: Hexokinase (most tissues) or Glucokinase (liver)
ATP used: 1 ATP
Product: Glucose-6-phosphate
Purpose: Trap glucose in the cell and commit it to metabolism
The Second Step of Glycolysis
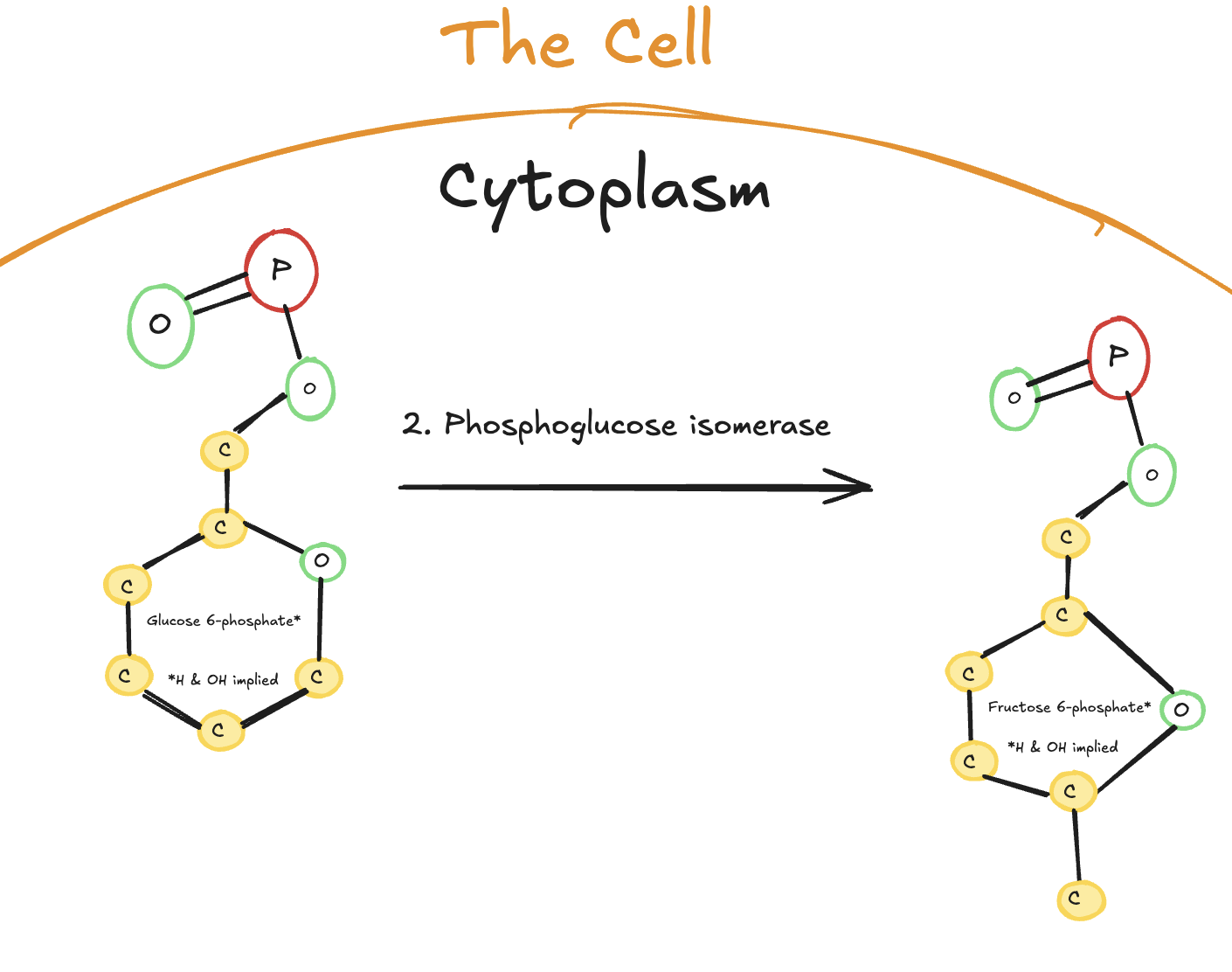
The second step of glycolysis is an isomerization reaction.
Digitally drawn diagram of the second step of glycolysis in the cytoplasm, showing glucose-6-phosphate converted into fructose-6-phosphate by phosphoglucose isomerase.
In this step, glucose-6-phosphate is converted into fructose-6-phosphate by the enzyme phosphoglucose isomerase. No ATP is used or produced here. Instead, the structure of the molecule is rearranged.
The key purpose of this step is preparation. Glucose-6-phosphate is a six-carbon ring with a functional group positioned in a way that is not ideal for later cleavage. By converting it into fructose-6-phosphate, the molecule is rearranged so that it can later be split evenly into two three-carbon molecules.
In other words, this step doesn’t extract energy. It sets up the molecule so that energy extraction becomes possible later in the pathway.
Key Takeaways
Enzyme: Phosphoglucose isomerase
Reaction type: Isomerization
ATP used or produced: None
Purpose: Rearrange the molecule to allow symmetrical splitting later
The Third Step of Glycolysis
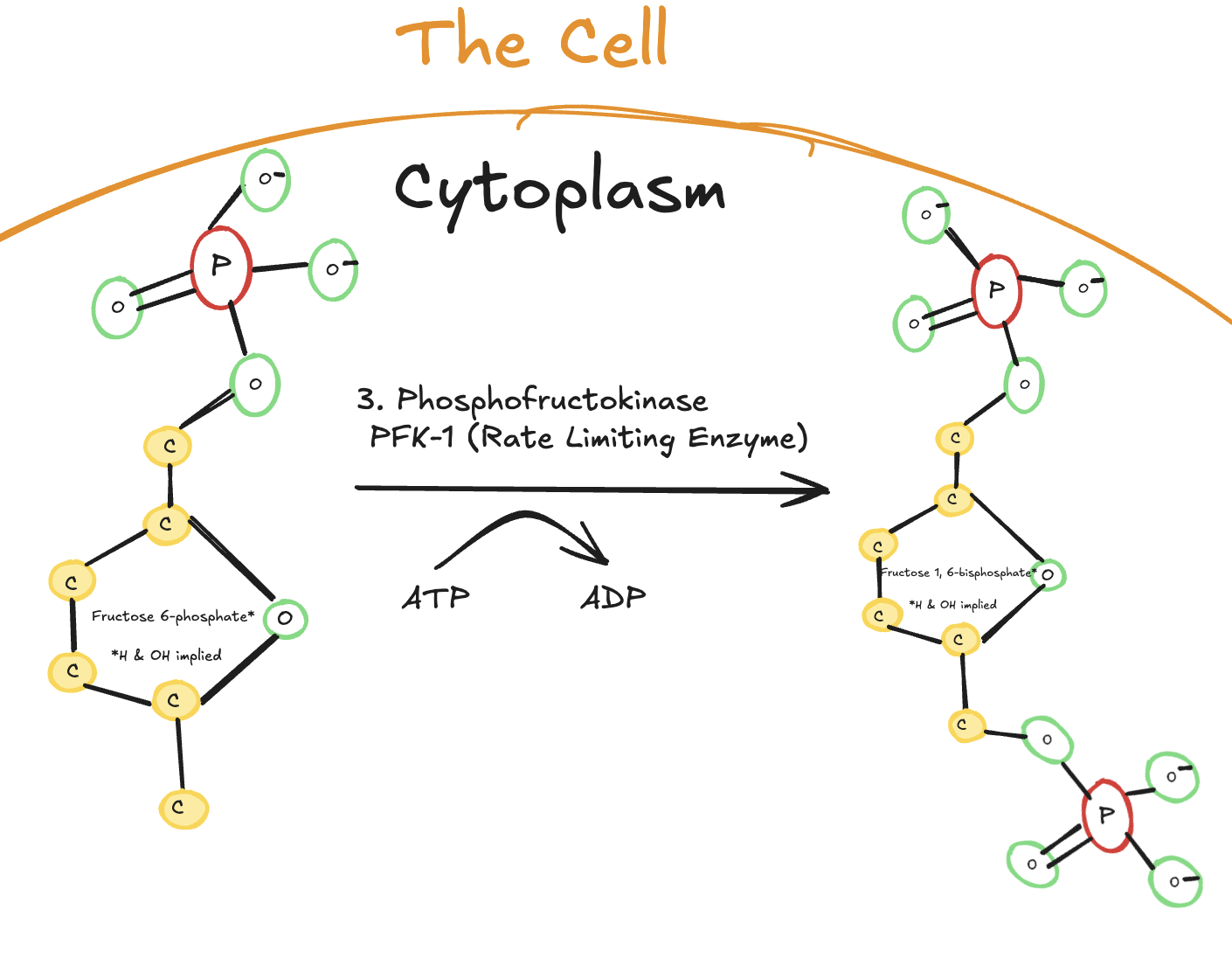
The third step of glycolysis is the phosphorylation of fructose-6-phosphate, and it is catalyzed by phosphofructokinase-1 (PFK-1).
Digitally drawn diagram of the third step of glycolysis in the cytoplasm, showing fructose-6-phosphate phosphorylated by phosphofructokinase-1 (PFK-1) using ATP to form fructose-1,6-bisphosphate.
The third step of glycolysis is the phosphorylation of fructose-6-phosphate, and it is catalyzed by phosphofructokinase-1 (PFK-1).
In this step, one molecule of ATP is used to add a second phosphate group to fructose-6-phosphate, producing fructose-1,6-bisphosphate. Like the first step of glycolysis, this reaction is irreversible under physiological conditions.
This step is especially important because it is the rate-limiting step of glycolysis. Once glucose-derived carbon passes this point, the cell is fully committed to completing glycolysis. Because of this, PFK-1 is tightly regulated and responds to the energy needs of the cell.
When energy is abundant, high levels of ATP inhibit PFK-1. When energy is low, molecules like AMP and ADP activate it. In this way, glycolysis speeds up when the cell needs ATP and slows down when energy demand is low.
Key Takeaways
Enzyme: Phosphofructokinase-1 (PFK-1)
Reaction type: Phosphorylation
ATP used: 1 ATP
Product: Fructose-1,6-bisphosphate
Reversibility: Irreversible
Significance: Rate-limiting and committed step of glycolysis
Regulation:
Inhibited by ATP
Activated by AMP and ADP
The Fourth Step of Glycolysis
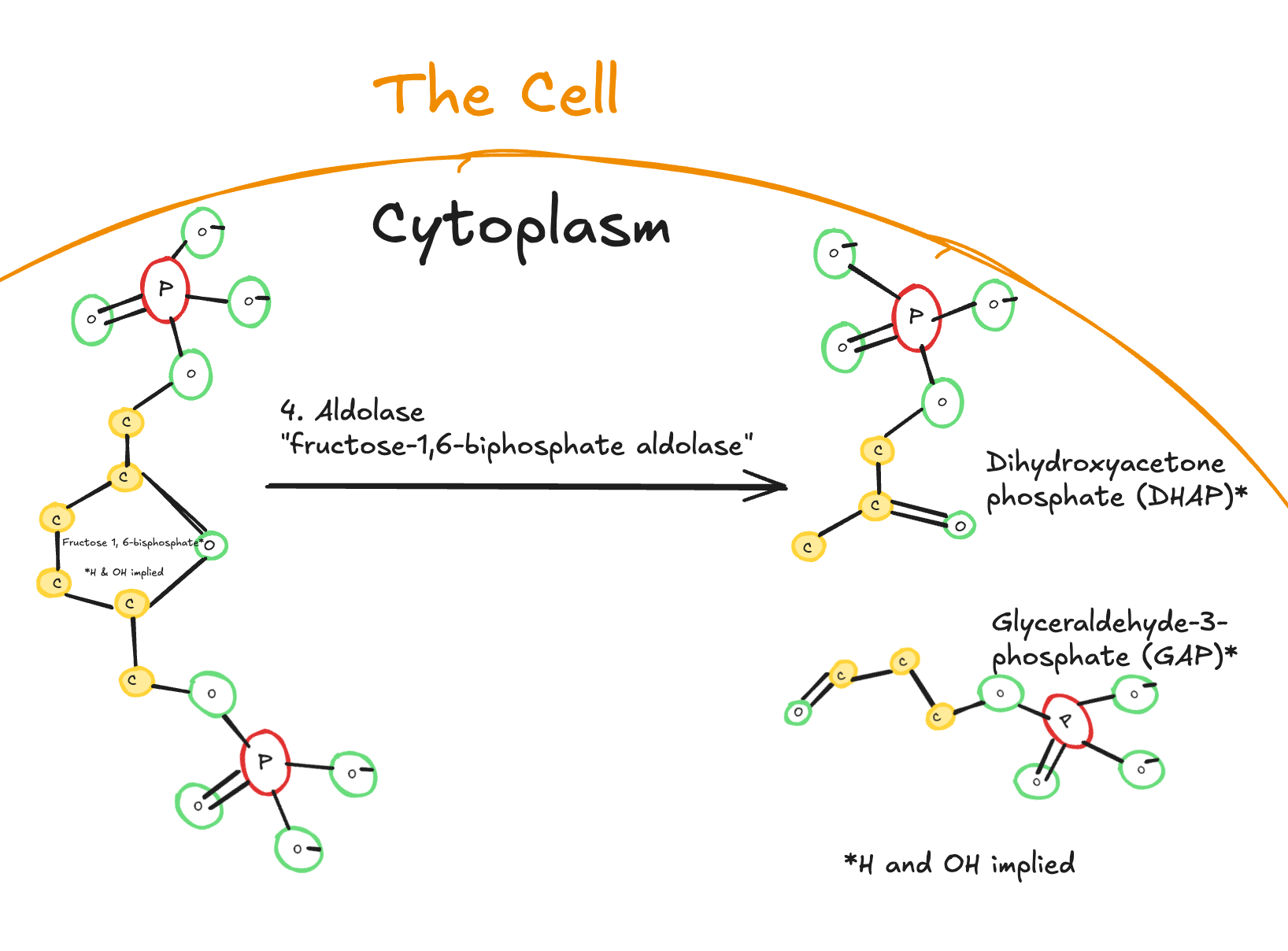
The fourth step of glycolysis is the cleavage of fructose-1,6-bisphosphate.
Digitally drawn diagram of the fourth step of glycolysis in the cytoplasm, showing fructose-1,6-bisphosphate cleaved by aldolase into dihydroxyacetone phosphate (DHAP) and glyceraldehyde-3-phosphate (GAP).
The fourth step of glycolysis is the cleavage of fructose-1,6-bisphosphate.
In this step, the enzyme aldolase splits the six-carbon molecule fructose-1,6-bisphosphate into two three-carbon molecules: dihydroxyacetone phosphate (DHAP) and glyceraldehyde-3-phosphate (GAP).
This is the first time in glycolysis that the carbon backbone is physically split. No ATP is used or produced here. Instead, the pathway transitions from a single six-carbon molecule to two three-carbon intermediates.
Key Takeaways
Enzyme: Aldolase
Reaction type: Cleavage
Substrate: Fructose-1,6-bisphosphate (6 carbons)
Products:
Dihydroxyacetone phosphate (DHAP)
Glyceraldehyde-3-phosphate (GAP)
ATP used or produced: None
Significance: Splits the molecule into two three-carbon intermediates
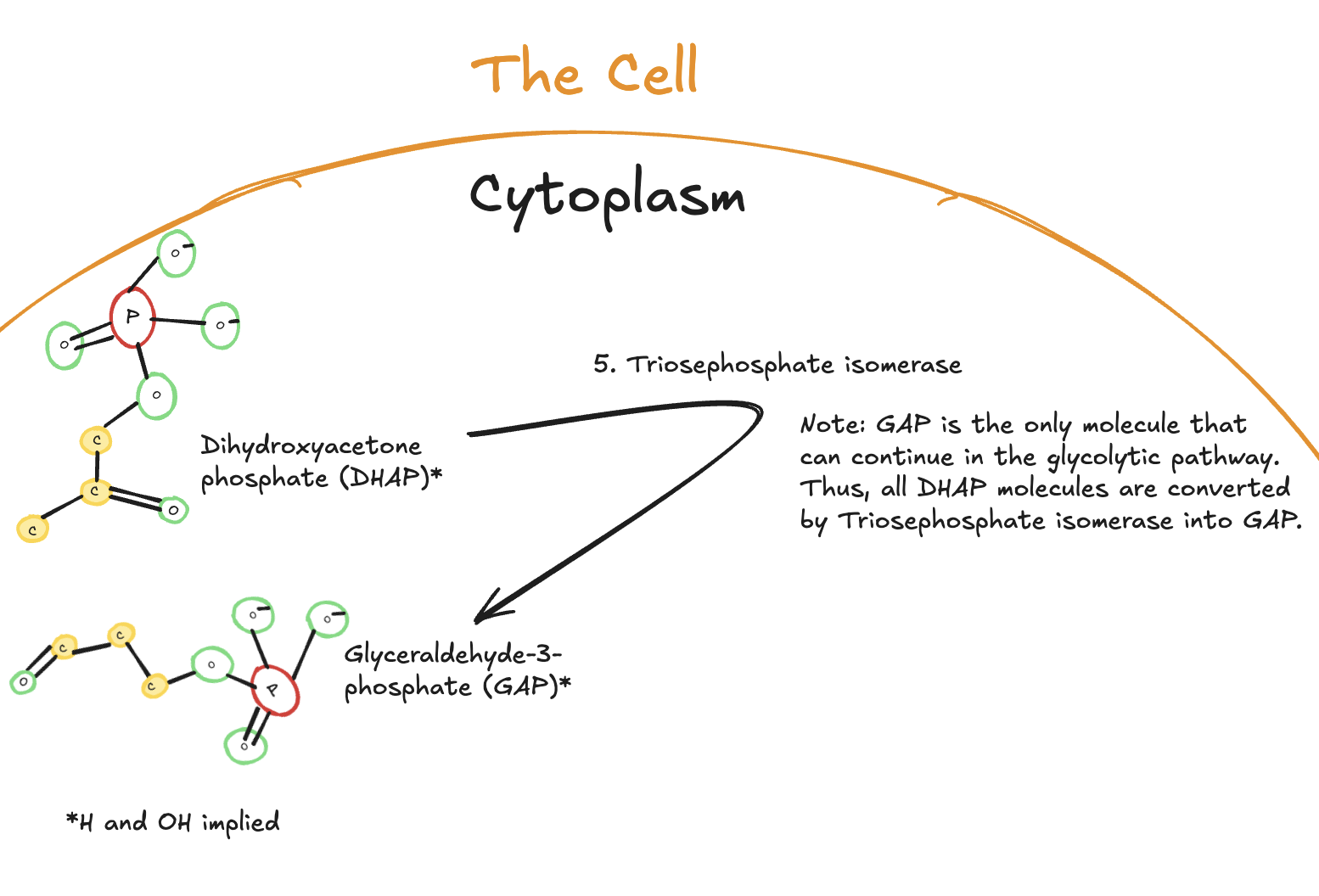
The Fifth Step of Glycolysis
The fifth step of glycolysis is an isomerization reaction catalyzed by triosephosphate isomerase.
Digitally drawn diagram of the fifth step of glycolysis in the cytoplasm, showing dihydroxyacetone phosphate (DHAP) converted into glyceraldehyde-3-phosphate (GAP) by triosephosphate isomerase.
In this step, dihydroxyacetone phosphate (DHAP) is converted into glyceraldehyde-3-phosphate (GAP). Although two three-carbon molecules were produced in the previous step, only GAP can continue through glycolysis.
As a result, all DHAP molecules are rapidly converted into GAP. From this point forward, every remaining step of glycolysis occurs twice per original glucose molecule, once for each GAP.
No ATP is used or produced during this step.
Key Takeaways
Enzyme: Triosephosphate isomerase
Reaction type: Isomerization
Substrate: DHAP
Product: Glyceraldehyde-3-phosphate (GAP)
ATP used or produced: None
Significance:
Ensures both three-carbon fragments continue through glycolysis
Marks the point where glycolysis proceeds in duplicate per glucose molecule
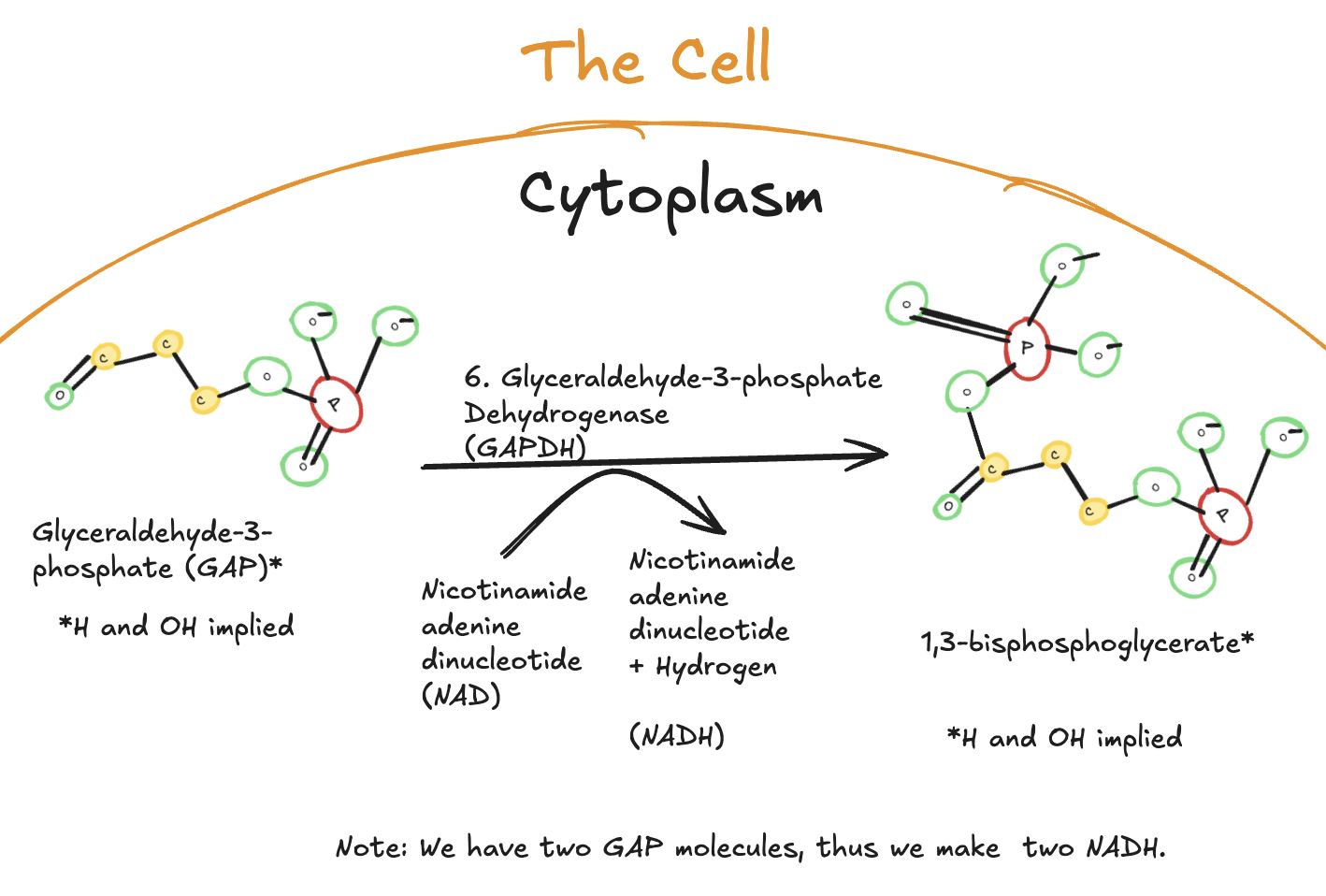
The Sixth Step of Glycolysis
The sixth step of glycolysis is catalyzed by glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and marks the beginning of the energy payoff phase.
Digitally drawn diagram of the sixth step of glycolysis in the cytoplasm, showing glyceraldehyde-3-phosphate oxidized to 1,3-bisphosphoglycerate by glyceraldehyde-3-phosphate dehydrogenase, with NAD⁺ reduced to NADH.
In this step, glyceraldehyde-3-phosphate (GAP) is oxidized and phosphorylated to form 1,3-bisphosphoglycerate. Importantly, this reaction reduces NAD⁺ to NADH, capturing high-energy electrons.
Because two GAP molecules are present per original glucose molecule, two NADH molecules are produced in this step.
No ATP is produced here, but this reaction is critical because it stores energy in the form of NADH, which can later be used for ATP production in the electron transport chain.
Key Takeaways
Enzyme: Glyceraldehyde-3-phosphate dehydrogenase (GAPDH)
Reaction type: Oxidation and phosphorylation
Substrate: Glyceraldehyde-3-phosphate (GAP)
Product: 1,3-bisphosphoglycerate
Electron carrier: NAD⁺ → NADH
Yield per glucose:
2 NADH
Significance:
First step that captures energy from glucose
Initiates the payoff phase of glycolysis
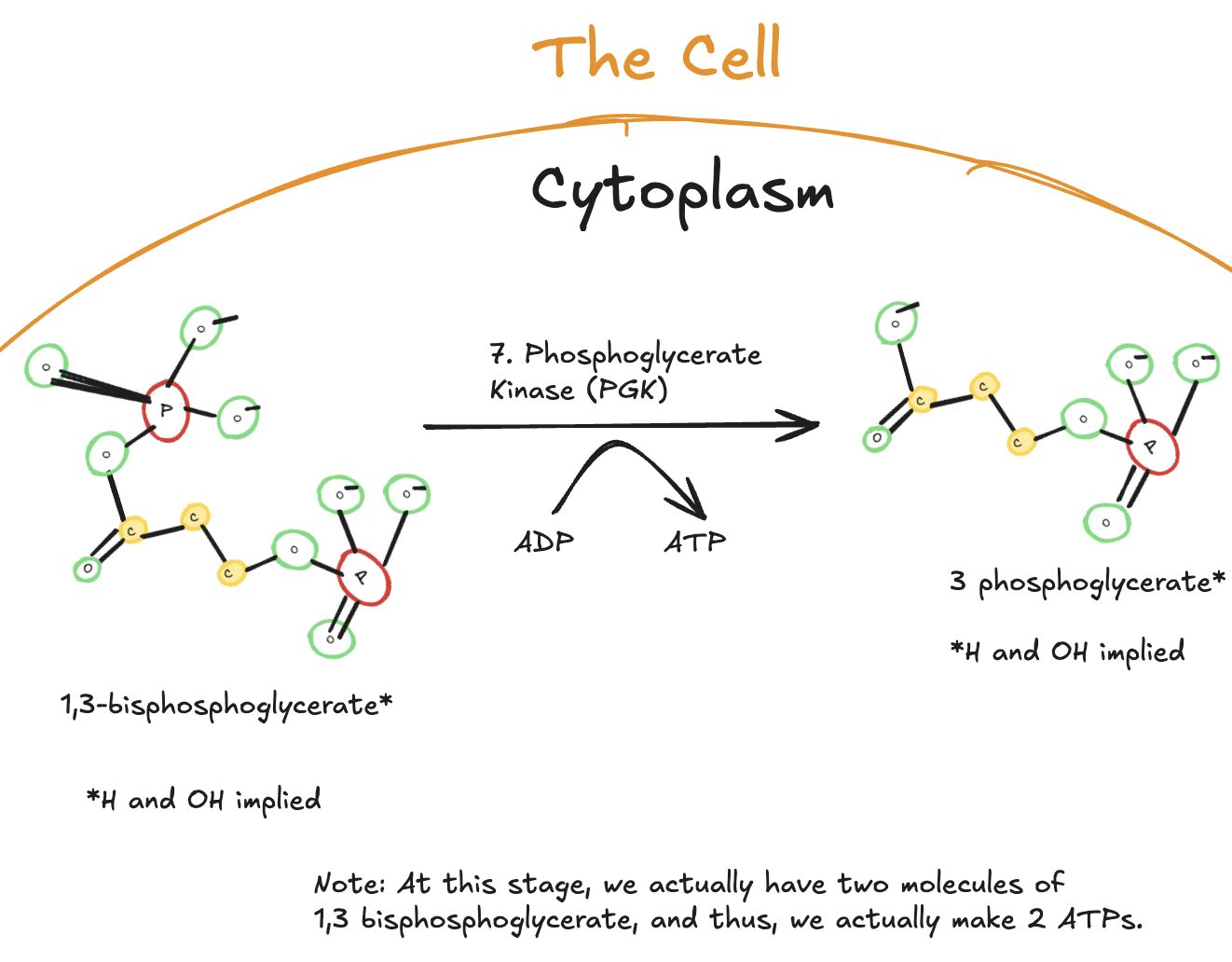
The Seventh Step of Glycolysis
The seventh step of glycolysis is the first ATP-producing reaction in the pathway.
Digitally drawn diagram of the seventh step of glycolysis in the cytoplasm, showing 1,3-bisphosphoglycerate converted to 3-phosphoglycerate by phosphoglycerate kinase, with ATP generated from ADP.
In this step, 1,3-bisphosphoglycerate donates a high-energy phosphate group to ADP, forming ATP. This reaction is catalyzed by phosphoglycerate kinase and produces 3-phosphoglycerate.
Because two molecules of 1,3-bisphosphoglycerate are present per original glucose molecule, two ATP molecules are generated at this step.
This reaction is an example of substrate-level phosphorylation, meaning ATP is produced directly from a metabolic intermediate rather than through the electron transport chain.
Key Takeaways
Enzyme: Phosphoglycerate kinase
Reaction type: Substrate-level phosphorylation
Substrate: 1,3-bisphosphoglycerate
Product: 3-phosphoglycerate
ATP yield per glucose:
2 ATP produced
Significance:
First ATP-generating step of glycolysis
Begins repayment of ATP invested earlier in the pathway
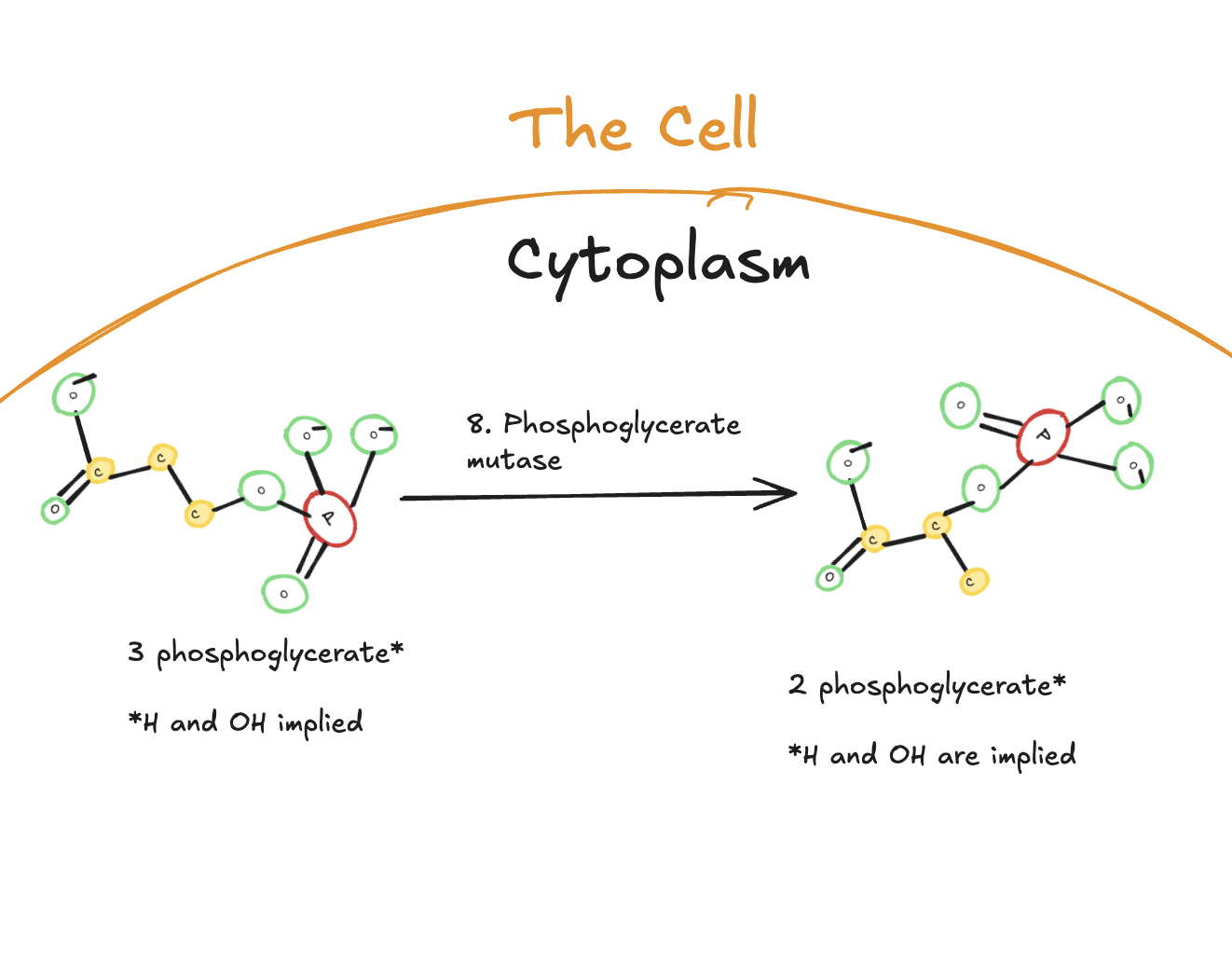
The Eighth Step of Glycolysis
The eighth step of glycolysis is a rearrangement reaction catalyzed by phosphoglycerate mutase.
Digitally drawn diagram of the eighth step of glycolysis in the cytoplasm, showing 3-phosphoglycerate converted to 2-phosphoglycerate by phosphoglycerate mutase.
In this step, 3-phosphoglycerate is converted into 2-phosphoglycerate. No ATP is used or produced. Instead, the phosphate group is shifted to a different position within the molecule.
The purpose of this step is preparation. By repositioning the phosphate group, the molecule is set up for the next reaction, which will create a high-energy intermediate capable of generating ATP.
Key Takeaways
Enzyme: Phosphoglycerate mutase
Reaction type: Intramolecular rearrangement
Substrate: 3-phosphoglycerate
Product: 2-phosphoglycerate
ATP used or produced: None
Significance:
Prepares the molecule for high-energy phosphate formation
Positions the phosphate group for ATP generation in the next step
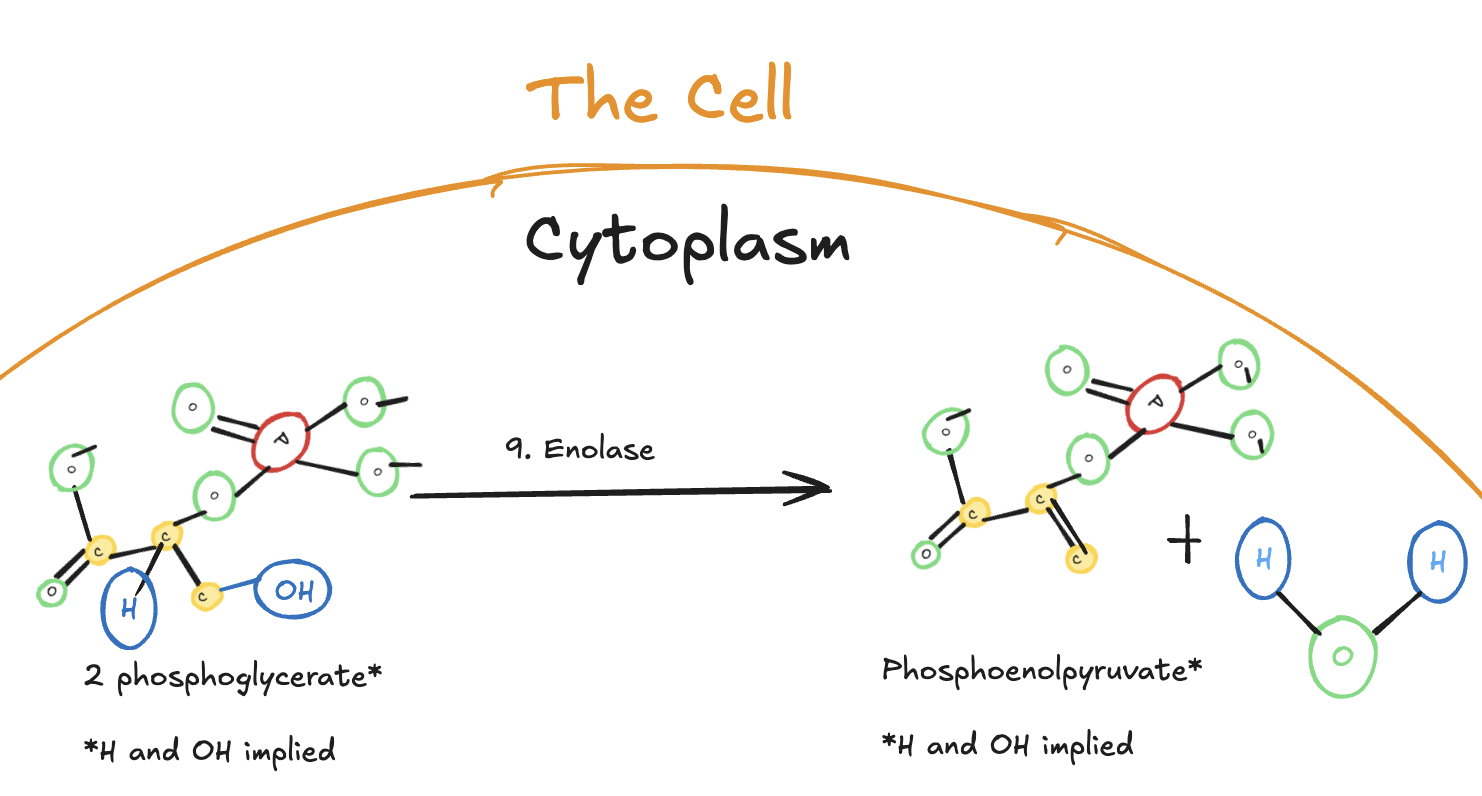
The Ninth Step of Glycolysis
The ninth step of glycolysis is a dehydration reaction catalyzed by the enzyme enolase.
Digitally drawn diagram of the ninth step of glycolysis in the cytoplasm, showing 2-phosphoglycerate converted to phosphoenolpyruvate (PEP) by enolase with the release of water.
In this step, 2-phosphoglycerate is converted into phosphoenolpyruvate (PEP) through the removal of a molecule of water. This dehydration reaction creates a very high-energy phosphate bond.
No ATP is produced at this step. Instead, the energy is stored in phosphoenolpyruvate, setting up the final step of glycolysis, where ATP will be generated.
Because two molecules of 2-phosphoglycerate are present per original glucose molecule, two molecules of phosphoenolpyruvate are formed.
Key Takeaways
Enzyme: Enolase
Reaction type: Dehydration
Substrate: 2-phosphoglycerate
Product: Phosphoenolpyruvate (PEP)
ATP used or produced: None
Yield per glucose:
2 PEP molecules
Significance:
Creates a high-energy phosphate intermediate
Prepares the pathway for the final ATP-generating step
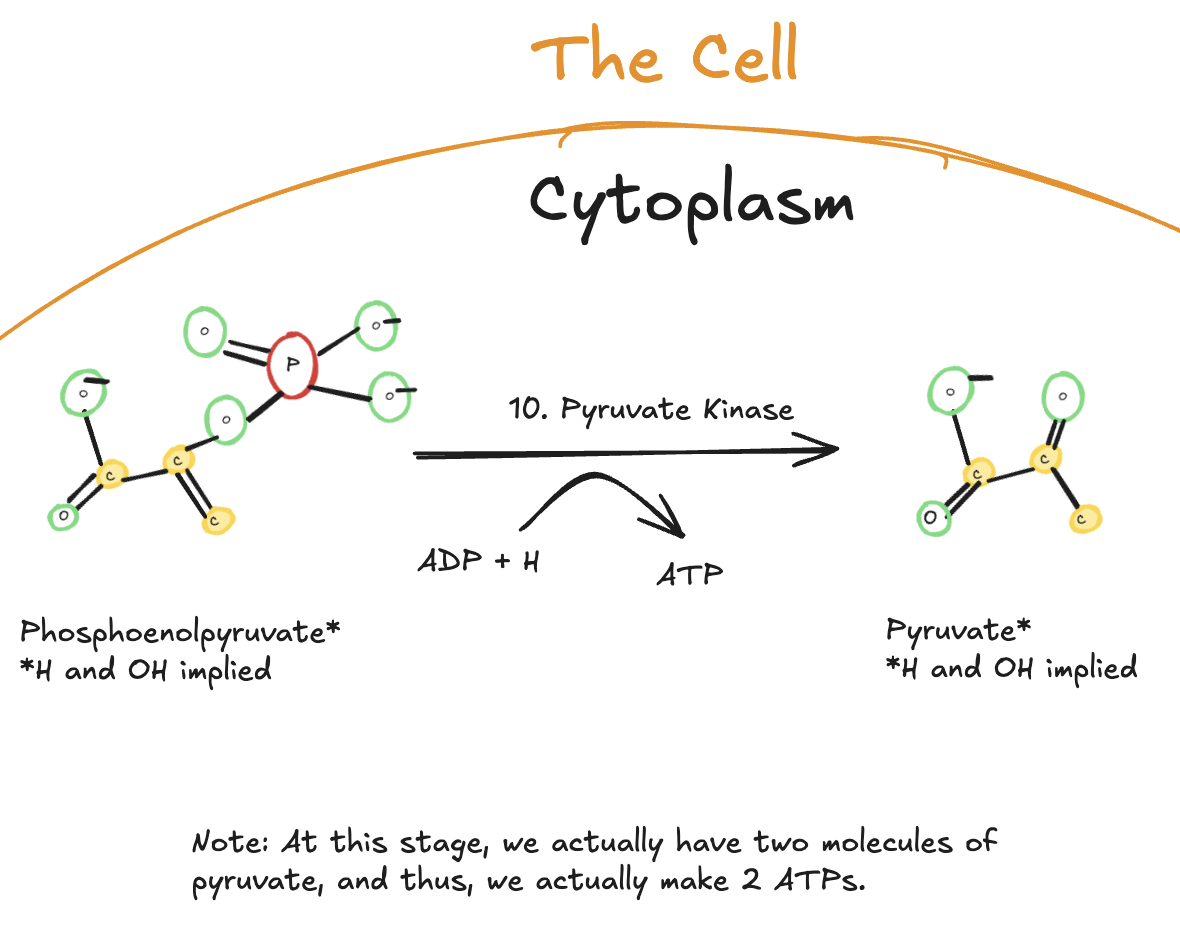
The Tenth Step of Glycolysis
The tenth and final step of glycolysis is catalyzed by pyruvate kinase.
Digitally drawn diagram of the tenth step of glycolysis in the cytoplasm, showing phosphoenolpyruvate (PEP) converted to pyruvate by pyruvate kinase, with ATP generated from ADP.
In this step, phosphoenolpyruvate (PEP) donates its high-energy phosphate group to ADP, producing ATP and forming pyruvate. This reaction is another example of substrate-level phosphorylation.
Because two molecules of PEP are present per original glucose molecule, two ATP molecules are generated in this step.
This reaction is irreversible and represents the final commitment of glucose-derived carbon to downstream metabolic pathways. The pyruvate produced can then enter the mitochondria for further oxidation or be reduced to lactate depending on oxygen availability and cellular demands.
Key Takeaways
Enzyme: Pyruvate kinase
Reaction type: Substrate-level phosphorylation
Substrate: Phosphoenolpyruvate (PEP)
Product: Pyruvate
ATP yield per glucose:
2 ATP produced
Reversibility: Irreversible
Significance:
Final ATP-producing step of glycolysis
Completes the conversion of glucose to pyruvate
Glycolysis: A Brief Summary
Glycolysis is the foundational pathway of cellular energy metabolism. It provides a rapid, oxygen-independent means of producing ATP and serves as the entry point for glucose-derived carbon into further metabolic pathways.
While the ATP yield of glycolysis alone is modest, its importance lies in speed, universality, and flexibility. Glycolysis can support immediate energy demands during high-intensity activity and supplies key intermediates for oxidative metabolism when oxygen is available.
From here, pyruvate represents a metabolic crossroads. Under sufficient oxygen, it can enter the mitochondria for further oxidation in the Krebs cycle. When energy demand outpaces oxygen availability, pyruvate can be reduced to lactate to sustain glycolytic ATP production.
Either way, glycolysis remains the starting point.
Key Points
Glycolysis converts one glucose molecule into two pyruvate molecules
Produces 2 ATP net via substrate-level phosphorylation
Generates 2 NADH, which carry high-energy electrons
Does not require oxygen
Occurs entirely in the cytoplasm
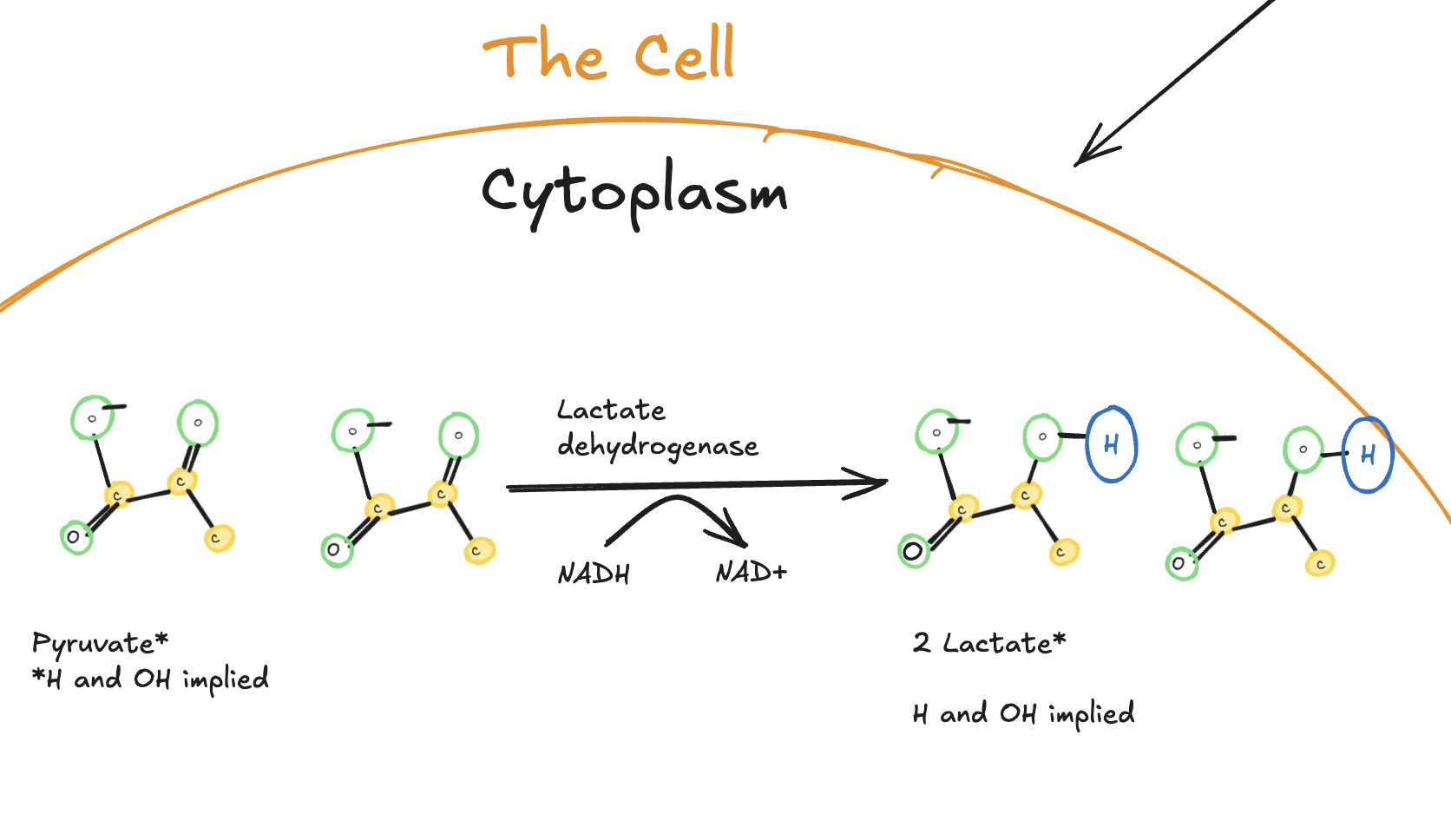
Lactate Fermentation
After glycolysis, a downstream reaction can occur during high-intensity exercise or when oxygen availability is limited.
What is it?
It’s basically a reduction from pyruvate to lactate.
Digitally drawn diagram showing pyruvate reduced to lactate in the cytoplasm by lactate dehydrogenase, with NADH oxidized to NAD⁺ to regenerate electron carriers, producing two lactate molecules per glucose.
In this process, pyruvate is reduced to lactate by the enzyme lactate dehydrogenase. At the same time, NADH is oxidized back to NAD⁺. This regeneration of NAD⁺ is critical, because it allows glycolysis to continue producing ATP when oxidative metabolism cannot keep up with energy demand.
Importantly, this reaction does not produce ATP itself. Its role is supportive. It sustains glycolysis by maintaining the availability of NAD⁺.
Key clarification
Lactate formation is not part of glycolysis
It occurs after glycolysis
It supports ATP production indirectly by preserving NAD⁺
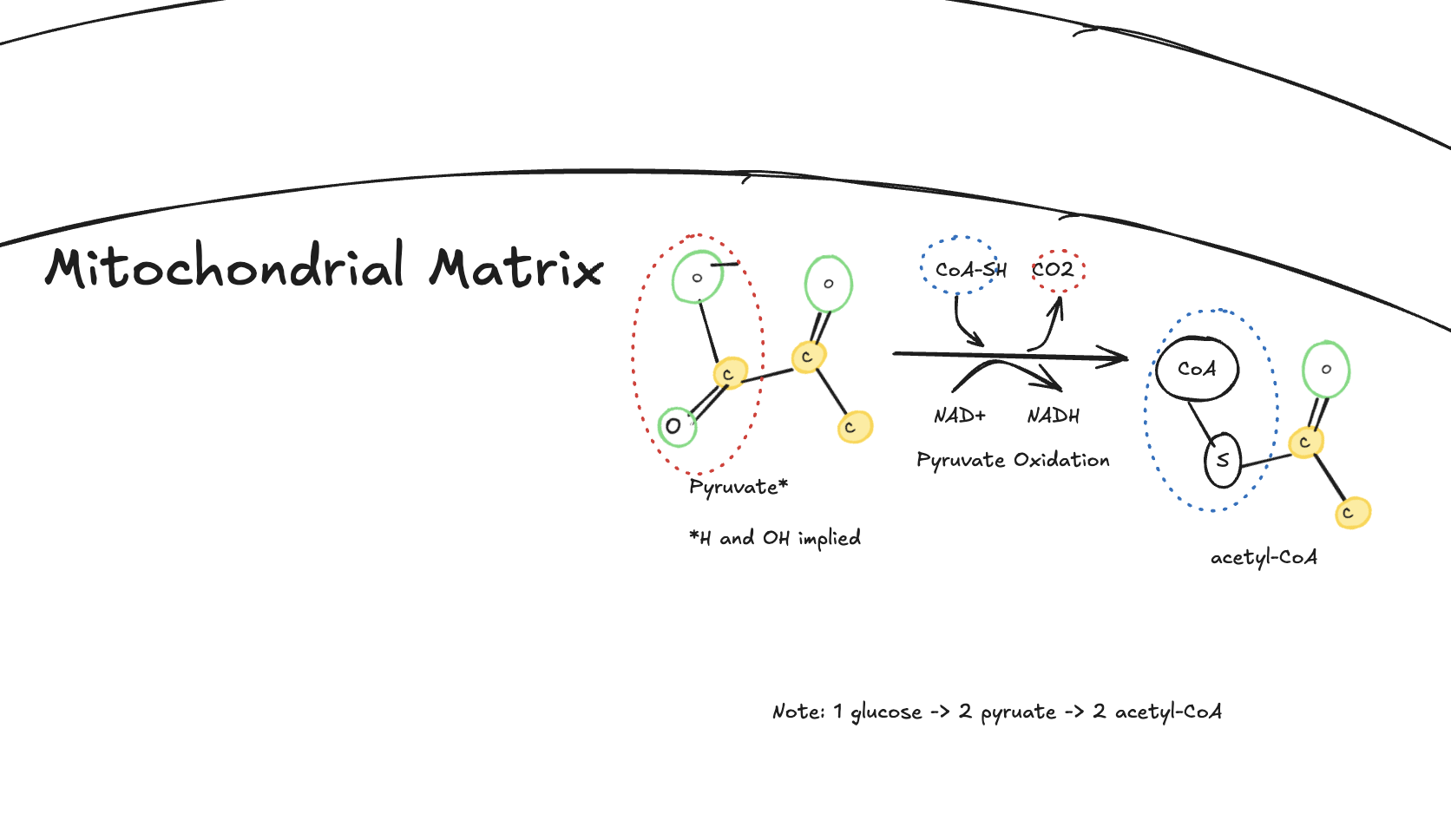
Pyruvate Oxidation
After glycolysis, pyruvate enters the mitochondria and undergoes pyruvate oxidation. Each pyruvate loses one carbon as CO₂, transfers electrons to NAD⁺ (forming NADH), and binds to coenzyme A to form acetyl-CoA.
Diagram illustrating pyruvate oxidation in the mitochondrial matrix, showing pyruvate converted into acetyl-CoA with the release of carbon dioxide and the reduction of NAD⁺ to NADH.
There, pyruvate is converted into acetyl-CoA by the pyruvate dehydrogenase complex. During this process:
One carbon is released as CO₂
NAD⁺ is reduced to NADH
Acetyl-CoA is formed and enters the Krebs cycle
Key takeaway:
This step links glycolysis to the Krebs cycle and commits carbon from glucose to aerobic metabolism.
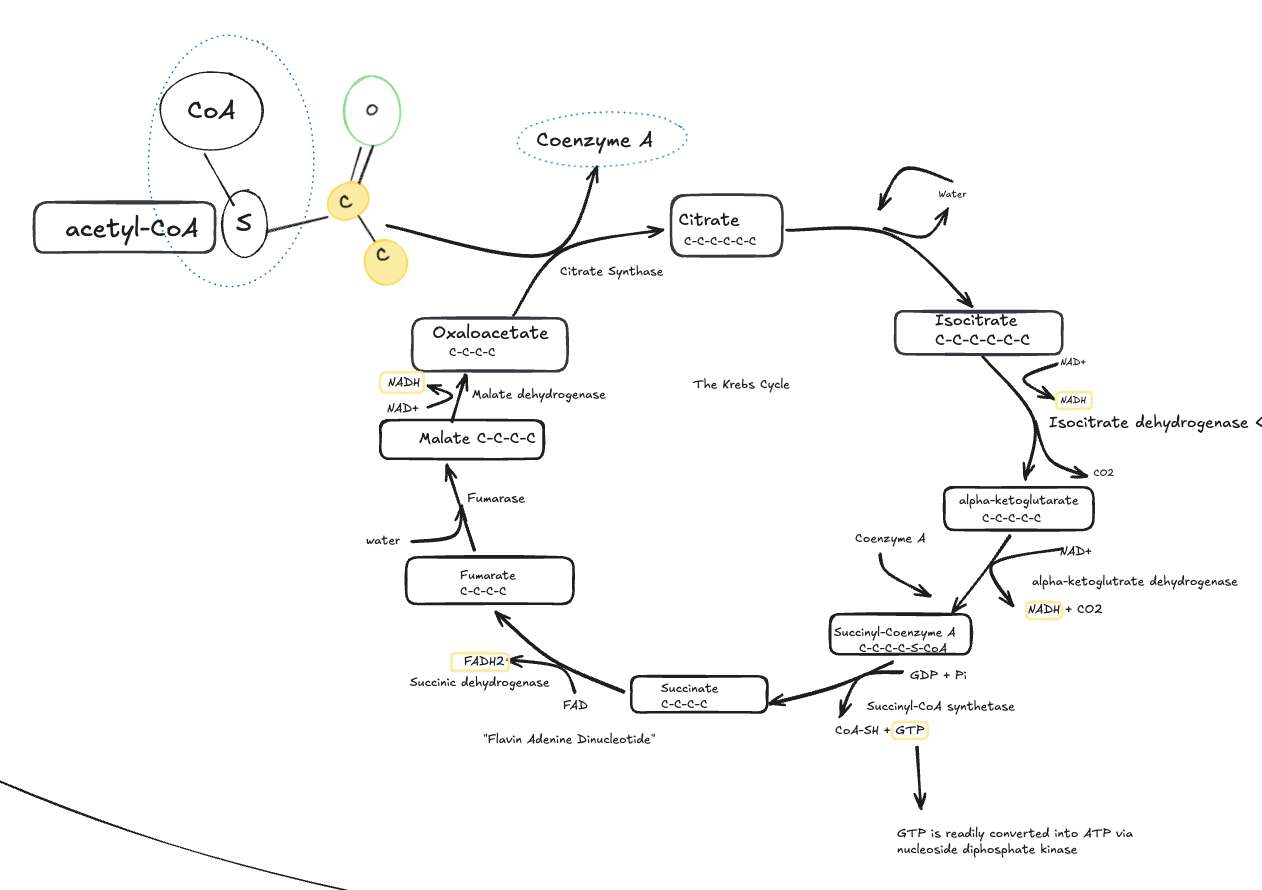
The Krebs Cycle (Citric Acid Cycle)
The Krebs cycle occurs in the mitochondrial matrix and fully oxidizes acetyl-CoA.
Hand-drawn schematic of the Krebs cycle in the mitochondrial matrix, showing acetyl-CoA entering the cycle, carbon dioxide release, and production of NADH, FADH₂, and GTP.
Acetyl-CoA combines with oxaloacetate to form citrate, initiating the Krebs cycle. Through a series of enzymatic reactions, carbons are released as CO₂ while high-energy electron carriers (NADH and FADH₂) are generated.
For each acetyl-CoA:
2 CO₂ are released
3 NADH are produced
1 FADH₂ is produced
1 GTP (≈ ATP) is generated
The Krebs cycle’s main role is electron harvesting, not ATP production.
Key takeaway:
The Krebs cycle does not directly produce much ATP, but it generates the reduced cofactors that power the electron transport chain.
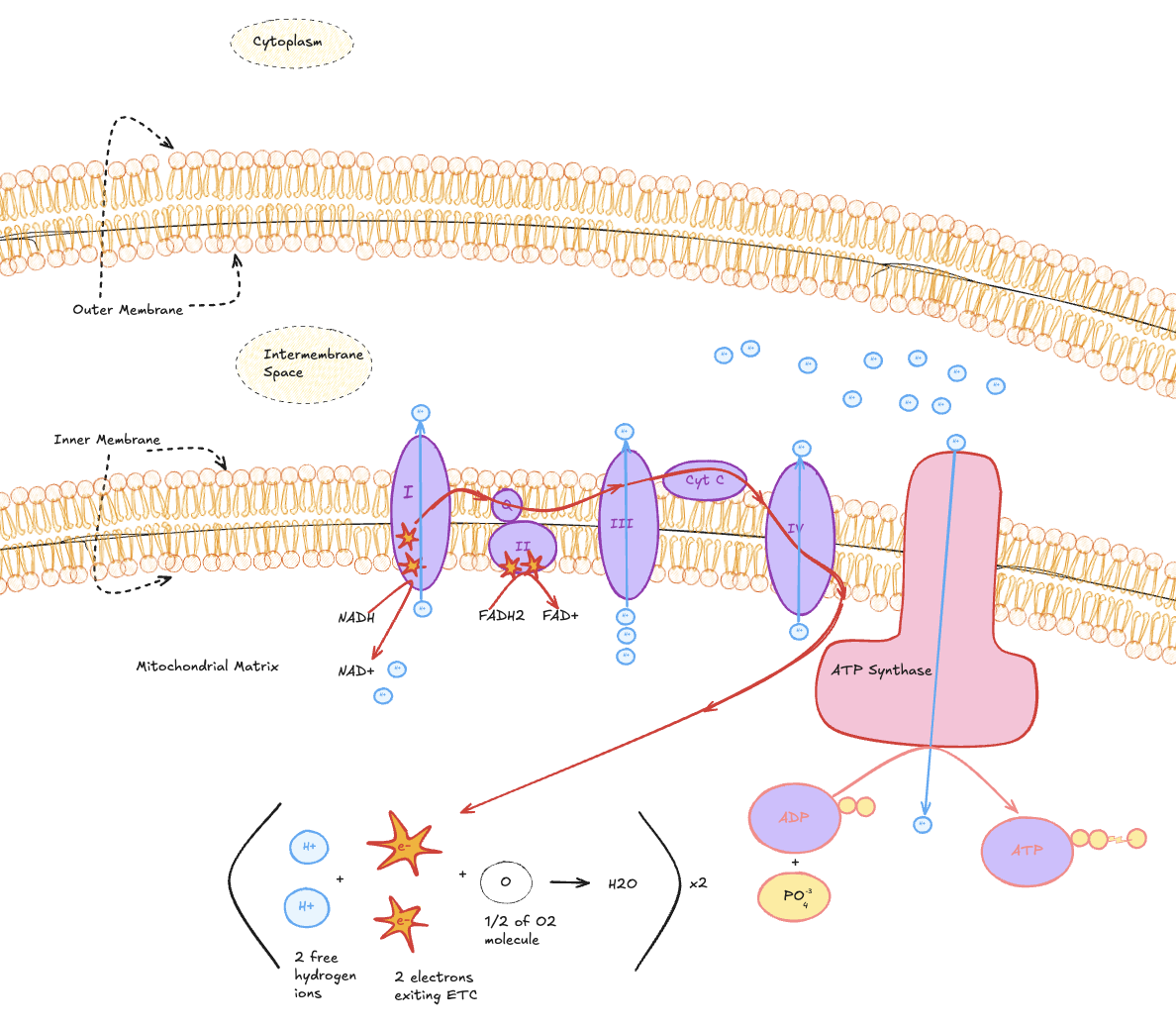
Electron Transport Chain (ETC)
The electron transport chain is located in the inner mitochondrial membrane.
Illustration of the electron transport chain across the inner mitochondrial membrane, showing complexes I–IV, proton pumping into the intermembrane space, ATP synthase activity, and oxygen as the final electron acceptor forming water.
Electrons from NADH and FADH₂ move through membrane-bound protein complexes, driving proton pumping and creating an electrochemical gradient. Protons flow back through ATP synthase, producing ATP. Oxygen accepts the electrons at the end, forming water.
The electron transport chain is located in the inner mitochondrial membrane.
NADH and FADH₂ donate electrons
Electron flow drives proton pumping
A proton gradient powers ATP synthase
Oxygen is the final electron acceptor, forming water
Key takeaway:
This is where the majority of ATP from glucose metabolism is produced, and why oxygen is essential for sustained aerobic energy production.
Together, glycolysis, pyruvate oxidation, the Krebs cycle, and the electron transport chain form a coordinated system that converts the chemical energy of glucose into ATP, with oxygen determining whether energy extraction can proceed efficiently or must rely on short-term adaptations.